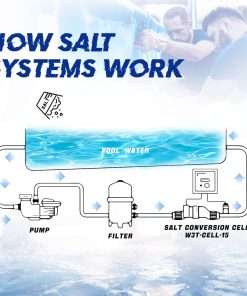
What is salt chlorination?
Salt chlorination is a cycle that utilizations salt to clean pool water. The salt is added to the water and afterward coursed through a generator that produces chlorine. The chlorine then kills microbes and different foreign substances in the water. Salt chlorination is a well known technique for pool sanitization since it is viable and generally simple to keep up with.
How is salt switched over completely to chlorine?
At the point when salt is added to water, it disintegrates and isolates into its part particles, sodium and chloride. These particles are then ready to lead power. In the event that an electric flow is gone through the pungent water, the sodium and chloride particles are drawn towards the terminals where they go through a cycle called electrolysis. At the positive terminal (the anode), chlorine gas is created. At the negative terminal (the cathode), hydrogen gas is created. The general synthetic response can be addressed as: 2 NaCl (aq) → 2 Na+ (aq) + Cl2 (g) + H2 (g) During electrolysis, the salt particles are separated into their part iotas. The chlorine particles are then delivered as gas, while the sodium and hydrogen iotas join to frame new atoms of water.
What is chlorine salt utilized for?
Chlorine salt is a typical synthetic compound that has a wide assortment of purposes. It is generally regularly utilized as a sanitizer or sanitizer, as it is powerful at killing microbes and other destructive microorganisms. It is additionally utilized in pools to assist with keeping the water perfect and liberated from pollutants. Likewise, chlorine salt can be utilized for cleaning, de-lubing, and carving.
What compound response is a salt chlorinator?
A salt chlorinator is a gadget that utilizes electrolysis to create chlorine from salt broke down in water. This interaction is known as electrolytic chlorination. The produced chlorine is utilized to sanitize pool water and guard it clean and for swimmers. Salt chlorinators are much of the time utilized in business and public pools since they are more compelling and effective than conventional chlorine generators. Salt chlorinators likewise have the additional advantage of being more straightforward to keep up with, since you just have to add salt to the water, instead of continually adding and observing chlorine levels. The substance response that happens in a salt chlorinator can be depicted as follows: When power is gone through salt water, the salt particles separate into sodium and chloride particles. These particles are drawn to the cathodes in the chlorinator, where they go through a substance response that produces chlorine gas. The created chlorine gas then breaks up into the pool water, where it goes about as a strong sanitizer. It kills microscopic organisms and different microorganisms, guarding the water clean and for swimming.